Net dipole moment11/4/2023 Bond dipole moments A diagram showing the bond dipole moments of boron trifluoride. The amount of covalent and ionic character depends on the values of the squared coefficients a 2 and b 2. Īs a quantum-mechanical description, Pauling proposed that the wave function for a polar molecule AB is a linear combination of wave functions for covalent and ionic molecules: ψ = aψ(A:B) + bψ(A +B −). He estimated that a difference of 1.7 corresponds to 50% ionic character, so that a greater difference corresponds to a bond which is predominantly ionic. .png) 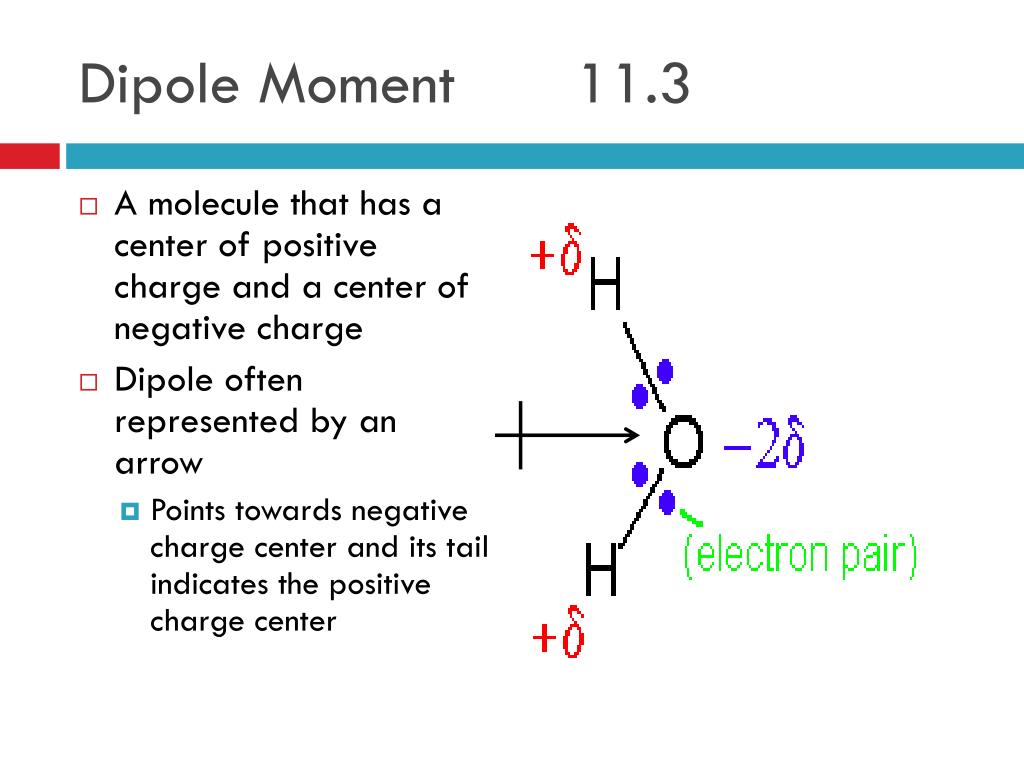
Pauling based this classification scheme on the partial ionic character of a bond, which is an approximate function of the difference in electronegativity between the two bonded atoms.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
